Biomarkers in disease diagnosis serve as measurable indicators of normal biological processes, pathogenic changes, or responses to therapeutic interventions. These biological signatures, which include proteins, nucleic acids, metabolites, and even cellular or imaging characteristics, enable laboratories to identify the presence of disease, assess its progression, predict outcomes, and monitor treatment effectiveness long before traditional symptoms become evident. In modern healthcare, biomarkers in disease diagnosis have transformed medicine from a reactive approach focused on overt clinical manifestations to a proactive strategy centered on early detection and personalized care. Laboratories play a central role in this shift by developing, validating, and applying these tools across a wide spectrum of conditions, from cancer and cardiovascular disease to neurological disorders and infectious illnesses.
The concept of biomarkers in disease diagnosis has roots in ancient observations, such as the use of urine color or odor to infer health status, but it gained scientific rigor in the 20th century with the identification of specific molecules like prostate-specific antigen for prostate cancer or cardiac troponin for myocardial injury. Today, advances in genomics, proteomics, and metabolomics have expanded the biomarker repertoire, allowing detection of subtle molecular alterations at the earliest stages of disease. For example, circulating tumor DNA in blood can signal the presence of cancer years before imaging reveals a mass, while elevated levels of phosphorylated tau in plasma help identify Alzheimer’s disease pathology before cognitive decline is apparent.
Laboratories detect biomarkers through a variety of methods, including immunoassays for proteins, polymerase chain reaction or next-generation sequencing for nucleic acids, and mass spectrometry for metabolites. These techniques require high sensitivity and specificity to distinguish pathological changes from normal variation or confounding factors like inflammation or medications. The clinical significance lies in their ability to stratify risk, guide therapy selection, and monitor response, ultimately improving survival rates and reducing unnecessary interventions.
This article examines the role of biomarkers in disease diagnosis, focusing on how laboratories utilize and identify them for early detection. It covers major categories of biomarkers, detection technologies, applications across disease areas, and challenges in implementation. A detailed section presents real data from recent studies and clinical validations between 2023 and 2025, highlighting performance metrics such as sensitivity, specificity, and predictive values. By understanding these tools, healthcare professionals can better integrate biomarkers in disease diagnosis into routine practice, enhancing precision and timeliness in patient care.
Categories and Mechanisms of Biomarkers in Disease Diagnosis
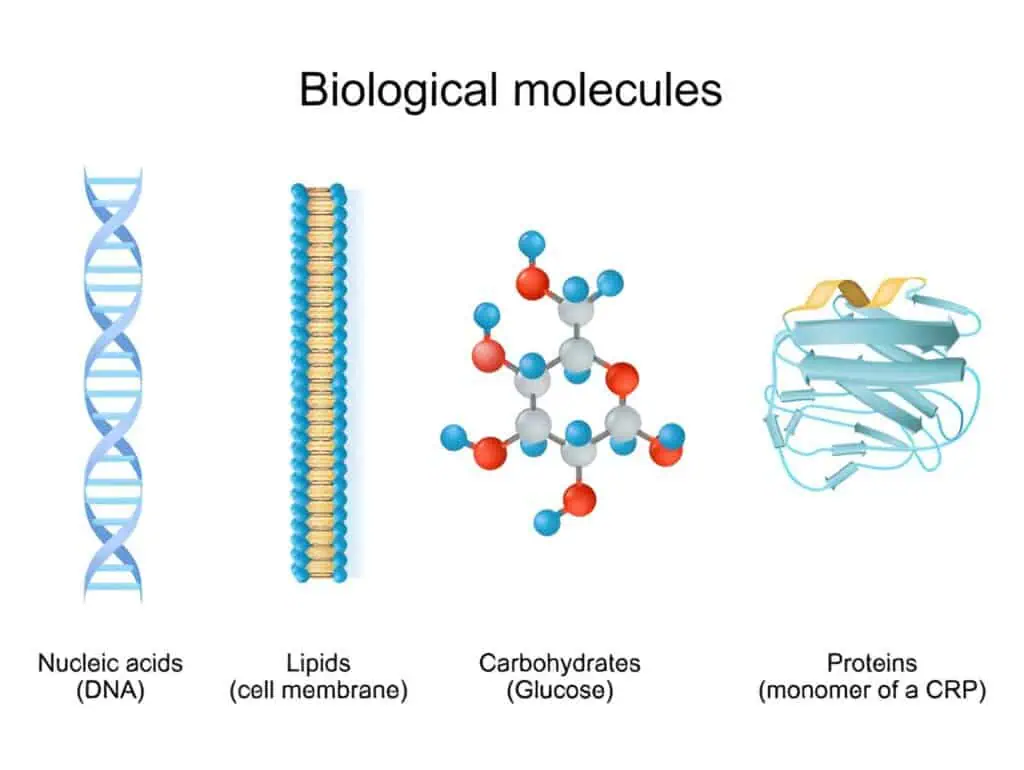
Biomarkers in disease diagnosis are broadly classified into diagnostic, prognostic, predictive, and monitoring types, each serving distinct clinical purposes. Diagnostic biomarkers confirm the presence of a disease or condition, often by detecting specific molecular signatures associated with pathological processes. Prognostic biomarkers provide information about the likely course of a disease, helping predict outcomes such as progression or survival. Predictive biomarkers identify individuals likely to respond to a particular therapy, supporting personalized treatment decisions. Monitoring biomarkers tracks disease status or treatment response over time.
At the molecular level, biomarkers in disease diagnosis include proteins, nucleic acids, metabolites, and cellular components. Protein biomarkers, such as cardiac troponin for myocardial infarction or prostate-specific antigen for prostate cancer, are detected through immunoassays that rely on antibody-antigen interactions. Nucleic acid biomarkers, including circulating tumor DNA or microRNAs, are analyzed via polymerase chain reaction or sequencing techniques that amplify and identify genetic alterations. Metabolite biomarkers, like elevated glucose or ketones in diabetes, reflect downstream biochemical changes and are measured using enzymatic assays or mass spectrometry.
Laboratories detect these biomarkers using a combination of traditional and advanced technologies. Enzyme-linked immunosorbent assays remain standard for protein quantification due to their high throughput and cost-effectiveness. For nucleic acids, digital droplet polymerase chain reaction offers absolute quantification with superior sensitivity for low-abundance targets. Next-generation sequencing enables comprehensive profiling of multiple mutations simultaneously, while mass spectrometry provides detailed metabolite and protein signatures. Emerging approaches, such as synthetic biomarkers that generate amplified signals in response to disease-specific enzymes, enhance detection limits for early-stage conditions.
The mechanisms underlying biomarker utility stem from their ability to reflect disease-specific biology. In cancer, for instance, tumor cells release circulating tumor DNA into the bloodstream, carrying mutations or methylation patterns unique to the malignancy. In cardiovascular disease, cardiac troponin release indicates myocardial injury, while in neurodegenerative disorders, phosphorylated tau or neurofilament light chain signals neuronal damage. Laboratories must validate these biomarkers against gold standards like imaging or biopsy to establish cutoffs that balance sensitivity and specificity.
The selection of biomarkers in disease diagnosis depends on the clinical context. Screening programs prioritize high specificity to minimize false positives, while diagnostic confirmation favors high sensitivity. Laboratories contribute by performing rigorous analytical validation, including precision, accuracy, and interference studies, to ensure reliable performance across diverse patient populations.
Applications of Biomarkers in Disease Diagnosis Across Major Conditions
Biomarkers in disease diagnosis find widespread application in oncology, cardiovascular medicine, neurology, and infectious diseases, enabling early intervention and tailored management. In oncology, prostate-specific antigen has long been used for prostate cancer screening, though its limitations in specificity have prompted the development of more refined panels incorporating free PSA ratios or prostate health index. For breast cancer, CA 15-3 and CA 27-29 assist in monitoring recurrence, while HER2 expression guides targeted therapy with trastuzumab. Liquid biopsy approaches using circulating tumor DNA have revolutionized early detection, identifying mutations in genes like EGFR or KRAS in lung and colorectal cancers with high concordance to tissue biopsy.
In cardiovascular disease, high-sensitivity cardiac troponin assays detect myocardial injury within hours of symptom onset, allowing rapid diagnosis of acute coronary syndromes. N-terminal pro-B-type natriuretic peptide serves as a prognostic marker in heart failure, with elevated levels predicting adverse events. C-reactive protein indicates systemic inflammation and improves risk stratification in atherosclerotic disease when combined with traditional factors.
Neurological applications include phosphorylated tau and amyloid-beta ratios for Alzheimer’s disease, where plasma p-tau217 has shown excellent accuracy in identifying amyloid pathology. Neurofilament light chain acts as a general marker of neuronal damage across multiple neurodegenerative conditions, aiding in differential diagnosis. In multiple sclerosis, the kappa free light chain index in cerebrospinal fluid enhances diagnostic specificity when used alongside oligoclonal bands.
For infectious diseases, biomarkers in disease diagnosis support rapid identification and monitoring. Procalcitonin distinguishes bacterial from viral infections, guiding antibiotic stewardship. In viral hepatitis, quantitative hepatitis B surface antigen or core-related antigen levels predict disease activity and treatment response. During pandemics, antigen or nucleic acid tests for pathogens like SARS-CoV-2 provide timely results for isolation and therapy decisions.
Laboratories integrate these biomarkers into multi-marker panels to improve overall performance. For example, combining cardiac troponin with natriuretic peptides enhances risk prediction in acute chest pain. In oncology, multi-cancer early detection tests using methylation patterns of circulating tumor DNA achieve high specificity while detecting multiple tumor types simultaneously.
The clinical significance of biomarkers in disease diagnosis extends to prognosis and therapy monitoring. Declining levels of tumor markers after treatment indicate response, while rising values signal recurrence. In chronic kidney disease, the estimated glomerular filtration rate combined with albuminuria biomarkers stages the disease and guides interventions.
Challenges and Future Directions in Biomarkers in Disease Diagnosis
Despite their promise, biomarkers in disease diagnosis face challenges related to validation, standardization, and accessibility. Many candidate biomarkers lack sufficient sensitivity or specificity for routine clinical use, particularly in early-stage disease where signal levels are low. Standardization across laboratories remains inconsistent, leading to variability in cutoffs and interpretations. Cost and infrastructure barriers limit adoption in low-resource settings, where advanced assays may not be feasible.
Regulatory hurdles also slow translation from discovery to practice. Rigorous analytical and clinical validation is required to demonstrate utility, often involving large prospective cohorts. Ethical considerations arise around incidental findings or overdiagnosis, which can cause anxiety or unnecessary procedures.
Future directions emphasize multi-omic integration and artificial intelligence to enhance performance. Combining genomic, proteomic, and metabolomic data into composite scores improves accuracy. Artificial intelligence algorithms analyze complex patterns, reducing false positives and enabling personalized thresholds. Liquid biopsy technologies, including extracellular vesicles and synthetic biomarkers, promise noninvasive detection with amplified signals for ultra-early diagnosis.
Point-of-care and wearable devices will democratize access, allowing continuous monitoring of dynamic biomarkers. Regulatory frameworks are evolving to support adaptive approvals for biomarker-driven therapies. As these advancements mature, biomarkers in disease diagnosis will increasingly shift healthcare toward prevention and precision, improving outcomes while reducing burdens on patients and systems.
Performance Metrics and Clinical Impact of Biomarkers in Disease Diagnosis

This section presents real data from studies and reports between 2023 and 2025 on the performance of biomarkers in disease diagnosis, including sensitivity, specificity, area under the curve values, and clinical outcomes. These metrics highlight the practical utility and limitations of key biomarkers across major disease areas.
For Alzheimer’s disease, plasma p-tau217 has demonstrated exceptional accuracy. A 2024 cohort study involving 786 participants reported area under the curve values of 0.92 to 0.96 for identifying elevated amyloid-beta pathology and 0.93 to 0.97 for tau pathology, comparable to cerebrospinal fluid biomarkers. In individuals with probable Alzheimer’s dementia, p-tau217 achieved positive predictive values above 95 percent across ages. In mild cognitive impairment, performance varied by age, with negative results ruling out amyloid pathology with negative predictive values of 90 to 99 percent in non-Alzheimer’s dementia syndromes. Another 2025 study confirmed plasma p-tau217/Aβ42 ratio with high concordance to amyloid PET, supporting its use as a triage tool that could reduce confirmatory testing by approximately 80 percent.
In oncology, liquid biopsy approaches using circulating tumor DNA show promising results for early detection. A 2025 multi-cancer early detection test based on cfDNA methylation achieved a specificity of 99.5 percent and an overall sensitivity of 51.5 percent, increasing with stage (approximately 17 percent in stage I, 40 percent in stage II, 77 percent in stage III, and 90 percent in stage IV). Cancer signal origin prediction accuracy reached 89 percent among true positives.
In a prospective interventional study, the test maintained high specificity above 99 percent and demonstrated feasibility for integration into clinical workflows. For prostate cancer, a three-biomarker urine RNA panel (TTC3, H4C5, EPCAM) yielded an area under the curve of 0.96, with sensitivity 94 percent and specificity 86 percent, outperforming PCA3 alone (area under the curve 0.83). It maintained high accuracy in PSA-negative cases and distinguished prostate cancer from benign conditions.
Cardiovascular biomarkers provide strong prognostic value. High-sensitivity cardiac troponin T and N-terminal pro-B-type natriuretic peptide levels predicted incident cancer risk in a multiethnic cohort free of baseline cardiovascular disease or cancer. Higher quartiles of both markers were associated with increased all-cancer incidence, with hazard ratios of 1.18 for hs-cTnT and 2.41 for NT-proBNP in fully adjusted models. For colorectal cancer, both markers showed significant associations, while NT-proBNP alone was linked to lung cancer risk.
In infectious and inflammatory conditions, procalcitonin and C-reactive protein guide antibiotic stewardship. Beta-D-glucan assays for invasive fungal infections reported sensitivity ranging from 69.9 to 100 percent and specificity from 73 to 97.3 percent for candidiasis, with an area under the curve of 0.85. Adjusted cutoffs improved sensitivity to 80 to 98.7 percent while maintaining 97.3 percent specificity across multiple fungal infections.
For multiple sclerosis, updated diagnostic criteria incorporate the central vein sign and paramagnetic rim lesions on MRI to enhance specificity. The kappa free light chain index in cerebrospinal fluid shows comparable sensitivity and specificity to oligoclonal bands, supporting its use as an alternative or complementary marker.
Market and adoption data reflect growing clinical integration. The global biomarkers market was valued at approximately 104.35 billion dollars in 2025, projected to reach 366.64 billion dollars by 2035 at a compound annual growth rate of 13.39 percent. The blood-based biomarkers segment for Alzheimer’s disease was estimated at 169.22 million dollars in 2025, expected to grow to 529.90 million dollars by 2033 at a 17.37 percent compound annual growth rate, driven by FDA-cleared p-tau217 tests.
These data, spanning cohorts of hundreds to millions, illustrate that biomarkers in disease diagnosis achieve sensitivities and specificities often exceeding 85 to 95 percent in validated settings, with liquid biopsy and plasma assays reducing invasive procedures while maintaining high negative predictive values for ruling out disease. Clinical implementation has led to earlier interventions, reduced unnecessary testing, and improved risk stratification, though performance varies by disease stage and population.
Conclusion
Biomarkers in disease diagnosis enable laboratories to detect early molecular and cellular changes that precede clinical symptoms, facilitating timely intervention and personalized management. From cardiac troponin in myocardial injury to p-tau217 in Alzheimer’s pathology and circulating tumor DNA in oncology, these tools have expanded diagnostic capabilities across medical fields. Laboratories detect and validate these biomarkers using immunoassays, sequencing, and mass spectrometry, ensuring high analytical performance through rigorous quality controls.
Real data confirm their clinical utility, with area under the curve values frequently above 0.90 and sensitivities/specificities in the 85 to 99 percent range for many established markers. Challenges such as standardization and accessibility persist, but ongoing advances in multi-omic integration and artificial intelligence promise to overcome them. As biomarkers in disease diagnosis continue to evolve, they will further empower laboratories to deliver precise, early insights that improve patient outcomes and transform healthcare delivery.