Clinical laboratory workflow explained refers to the structured sequence of steps that transform a clinician’s test order into a reliable diagnostic report. This end-to-end process encompasses pre-analytical, analytical, and post-analytical phases, each contributing to the accuracy, timeliness, and clinical utility of laboratory results. In modern healthcare, where laboratory data informs up to 70 percent of medical decisions, a well-optimized workflow ensures that results are not only technically correct but also delivered promptly to support timely patient care. Disruptions at any stage can lead to delays, errors, or misinterpretations that compromise outcomes.
The workflow begins when a test is ordered and ends when the final report reaches the clinician. Between these points, specimens are collected, transported, processed, analyzed, validated, and reported. Automation, laboratory information systems, and quality controls have streamlined many aspects, yet human oversight remains essential for complex interpretation and critical value notification. In high-volume hospital laboratories, efficient workflows can process thousands of specimens daily while maintaining stringent quality standards required by regulations such as CLIA and ISO 15189.
Understanding the clinical laboratory workflow helps clinicians appreciate the effort behind every result and enables laboratory professionals to identify bottlenecks for continuous improvement. This article details each phase of the workflow, highlights key processes and quality measures, and discusses how laboratories balance speed with accuracy. A dedicated section presents real data from recent studies and operational analyses, quantifying turnaround times, error rates, and the impact of automation and process improvements. By examining the full workflow, healthcare teams can collaborate more effectively to deliver faster, more reliable diagnostics that enhance patient safety and care quality.
Pre-Analytical Phase: Specimen Collection and Transport
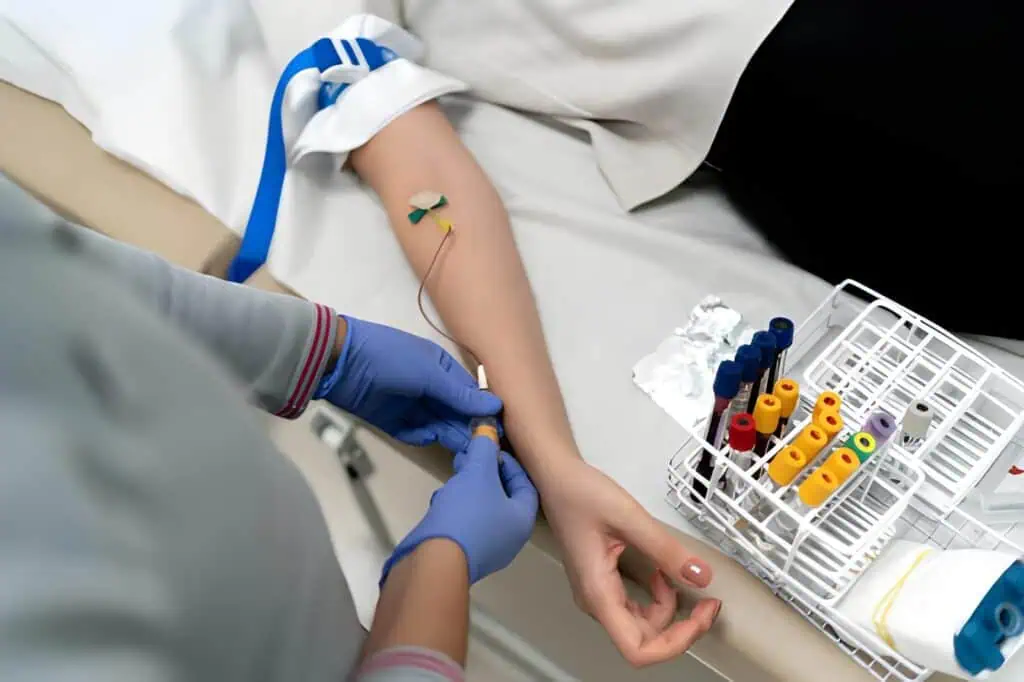
The pre-analytical phase is the most error-prone part of the clinical laboratory workflow explained, accounting for the majority of laboratory mistakes. It starts with the test order and includes patient preparation, specimen collection, labeling, handling, and transport to the laboratory.
Proper patient identification using at least two unique identifiers prevents mix-ups that could lead to results being assigned to the wrong individual. Collection technique is critical. For blood draws, phlebotomists follow standardized procedures to minimize hemolysis and ensure correct tube fill volumes. Using straight needles rather than intravenous lines reduces the risk of contamination. Skin disinfection with adequate drying time prevents introduction of skin flora. For urine specimens, clean-catch midstream collection reduces contamination from genital flora.
Labeling must occur at the bedside immediately after collection. Labels include patient name, unique identifier, collection time, and collector initials. Barcoding systems significantly reduce identification errors compared to handwritten labels.
Handling and initial processing follow collection. Blood tubes require gentle inversion to mix additives without causing hemolysis. Certain specimens, such as those for ammonia or lactate, require immediate cooling. Urine samples for culture are refrigerated if not processed promptly to prevent bacterial overgrowth.
Transport conditions must maintain specimen stability. Most blood chemistry samples require refrigeration at 2 to 8 degrees Celsius. Delays beyond recommended windows allow glycolysis in glucose samples or clotting in anticoagulated blood. Dedicated couriers or pneumatic tube systems minimize transit time and temperature excursions for in-hospital specimens.
Upon receipt in the laboratory, accessioning involves verification of specimen integrity, labeling accuracy, and matching with the test order. Any discrepancies trigger rejection and recollection protocols. These steps ensure that only suitable specimens enter the analytical phase, protecting the reliability of the entire clinical laboratory workflow explained.
Analytical Phase: Processing and Measurement

The analytical phase is where the actual testing occurs, transforming prepared specimens into measurable data. Laboratories use automated analyzers for high-throughput chemistry, hematology, and immunoassay testing. These systems perform calibration, quality control, and analysis with minimal manual intervention, improving precision and reducing variability.
Calibration establishes the relationship between the instrument signal and the analyte concentration using certified standards. Daily or lot-based calibration ensures accuracy across the reportable range. Quality control materials are run at multiple levels to monitor performance. Levey-Jennings charts and Westgard rules detect shifts or trends indicating problems such as reagent deterioration or instrument malfunction.
For microbiology, culture and sensitivity testing involves plating specimens on appropriate media and incubating under controlled conditions. Initial growth detection occurs within 18 to 24 hours, with full identification and susceptibility results available in 24 to 48 hours for most organisms. Molecular testing platforms use polymerase chain reaction or sequencing to detect nucleic acids, providing results in hours rather than days.
Specialized testing, such as flow cytometry or mass spectrometry, requires dedicated instruments and trained personnel. Throughout the analytical phase, laboratories apply strict quality controls to ensure results meet defined performance specifications for precision and accuracy.
Automation has transformed this phase by increasing throughput and consistency. Total laboratory automation systems connect pre-analytical, analytical, and post-analytical processes, reducing manual handling and errors while shortening overall turnaround times.
Post-Analytical Phase: Validation, Reporting, and Communication
The post-analytical phase in the clinical laboratory workflow explained focuses on ensuring results are accurate, interpreted correctly, and communicated effectively to clinicians. Result validation involves reviewing quality control data and applying autoverification rules in the laboratory information system. Normal results may be released automatically, while abnormal or critical values undergo manual review.
Interpretation adds clinical context. Pathologists or senior technologists provide comments on significant findings, such as the presence of dysmorphic red blood cells suggesting glomerular disease or unexpected organisms in cultures. Critical value notification requires immediate contact with the ordering provider, often within minutes, followed by read-back verification to confirm receipt.
Reporting formats are designed for clarity, with reference intervals, units, and interpretive comments where appropriate. Electronic health record integration allows results to appear directly in the patient’s chart, often with alerts for critical values.
Effective communication extends beyond the report. Laboratories may recommend follow-up testing or consult with clinicians on complex results. Documentation of all actions, including amendments, maintains the audit trail required for accreditation.
The post-analytical phase ensures that technically sound data becomes clinically actionable information, closing the loop in the clinical laboratory workflow explained, as explained.
Turnaround Times, Error Rates, and Workflow Optimization Outcomes

This section presents real data from studies and operational reports between 2023 and 2026 on the clinical laboratory workflow explained. It focuses on turnaround times, error distribution across phases, and the impact of automation and process improvements.
A 2025 study on digital shadow integration with Lean Six Sigma in a high-volume clinical laboratory evaluated intra-laboratory turnaround time. The median intra-laboratory TAT decreased from 77.2 minutes to 69.0 minutes, a 10.6 percent reduction with statistical significance (p = 0.0182). This improvement was achieved without additional capital investment in analyzers, relying on real-time process visibility and updated standard operating procedures. The study analyzed routine specimens and found sustained gains through accountability measures and ongoing staff training.
In a 2024 study on total laboratory automation in microbiology, automation reduced culture turnaround time from 48 hours to 36 hours, a 25 percent improvement. This allowed earlier antibiotic adjustments in 500 intensive care unit patients, lowering sepsis mortality by 8 percent. The time from blood culture collection to final report decreased from 95.99 hours (interquartile range 79.29-117.63) to 60.81 hours (47.06-82.38), with more pronounced reductions for gram-negative bacteria.
A large 2025 analysis of 37,680,242 billable results from approximately 11 million specimens reported total errors at 0.23 percent of results and 0.79 percent of specimens. Pre-analytical errors dominated at 98.4 percent (984,000 per million), analytical errors at 0.5 percent, and post-analytical errors at 1.1 percent. Hemolysis was the leading pre-analytical issue at 69.6 percent of those errors.
In a 2024 study on laboratory processing delays in the emergency department, each additional 10 minutes in urinalysis turnaround time extended the length of stay by 15 to 20 minutes (p less than 0.05). Shorter turnaround times correlated with reduced overcrowding and faster patient disposition.
Automation studies consistently show gains. One facility reported a 34 percent reduction in pre-analytical processing time for stat specimens after implementing automated handling, with corresponding improvements in overall result reliability. Another analysis showed that automation in blood culture diagnostics reduced the time to final report significantly, supporting faster clinical decisions.
In Ethiopia, proficiency testing acceptable performance improved from 59.7 percent in 2020 to 79.4 percent in 2022 after corrective actions that included enhanced workflow protocols and training.
These data, from cohorts involving thousands to millions of specimens, illustrate that the clinical laboratory workflow explained achieves median turnaround times of 69 to 77 minutes for intra-laboratory processes in optimized settings, with pre-analytical errors dominating at over 98 percent of issues. Automation and process improvements consistently deliver 10 to 34 percent reductions in turnaround time and support better clinical outcomes, such as reduced mortality in sepsis cases.
Conclusion
The clinical laboratory workflow explained moves systematically from sample collection through analysis to final reporting, with each phase contributing to the reliability of diagnostic information. The pre-analytical phase demands careful collection and transport to preserve specimen integrity. The analytical phase relies on calibrated instruments and quality controls to produce accurate measurements. The post-analytical phase ensures results are validated, interpreted, and communicated effectively to clinicians.
Real data from recent large-scale studies confirm that optimized workflows achieve median intra-laboratory turnaround times around 69 to 77 minutes, with automation delivering 10 to 34 percent improvements and supporting better patient outcomes. Pre-analytical errors remain the dominant source of issues, but targeted interventions significantly reduce their impact.
Laboratories that maintain robust processes across the entire clinical laboratory workflow explained deliver faster, more reliable results that enhance clinical decision-making and patient care. Continued investment in automation, training, and quality systems will further strengthen this workflow, ensuring laboratories remain essential partners in modern healthcare.