Specimen transport in clinical laboratories is a critical link in the diagnostic chain, bridging the gap between patient sample collection and laboratory analysis. Proper transport ensures that biological specimens, such as blood, urine, tissue, or swabs, arrive at the testing facility in a condition that preserves their integrity for accurate results. Delays, temperature excursions, or improper packaging can degrade analytes, introduce contamination, or render samples unusable, leading to recollection, delayed diagnoses, and increased healthcare costs. In high-volume labs processing thousands of specimens daily, even small inefficiencies in transport can cascade into significant operational challenges and patient harm.
The process involves multiple stakeholders, including phlebotomists, couriers, and lab personnel, each responsible for maintaining the chain of custody, temperature control, and regulatory compliance. Standards from the Clinical and Laboratory Standards Institute (CLSI) and the International Organization for Standardization (ISO 20658) outline requirements for collection and transport to minimize pre-analytical errors, which account for up to 70 percent of total laboratory mistakes. In 2026, with growing reliance on point-of-care testing and decentralized care, robust transport systems are essential for reliability, especially in remote or resource-limited settings where specimens may travel long distances.
This article examines best practices for specimen transport in clinical laboratories, focusing on packaging, temperature management, time limits, labeling, and safety protocols. It addresses common challenges like hemolysis in blood samples or bacterial overgrowth in urine and offers practical strategies for mitigation. A detailed section presents real data from studies and regulatory reports between 2020 and 2025, quantifying error rates, the effects of delays, and the benefits of optimized systems. By implementing these practices, laboratories can enhance diagnostic accuracy, protect personnel and the public from biohazards, and support timely patient care.
Fundamentals of Specimen Transport
Specimen transport begins immediately after collection and requires careful planning to maintain sample stability. The primary goal is to prevent changes in the specimen that could affect test results, such as glycolysis in glucose samples or clotting in anticoagulated blood. Key principles include using appropriate containers, maintaining required temperatures, and ensuring timely delivery.
Containers must be leak-proof, sterile where needed, and compatible with the specimen type. For blood, evacuated tubes with additives like EDTA or heparin preserve cellular components or plasma. Urine specimens often use preservative tubes or refrigeration to inhibit bacterial growth. Tissue samples require fixation or transport media to prevent autolysis. Secondary packaging, such as sealed bags with absorbent material, provides an additional barrier against leaks.
Temperature control is paramount. Most blood chemistry samples require 2 to 8 degrees Celsius during transport to slow metabolic processes. Frozen specimens, like those for certain molecular tests, need dry ice or specialized coolers to remain below minus 20 degrees Celsius. Room-temperature transport suits some microbiology swabs if delivered quickly. Exceeding these ranges can cause hemolysis, falsely elevating potassium levels by 20 to 30 percent, or degrade enzymes, leading to inaccurate liver function tests.
Time limits vary by analyte. CLSI guidelines recommend processing blood within 2 hours for most chemistry tests to avoid artifactual changes. Urine for culture should reach the lab within 2 hours at room temperature or 24 hours if refrigerated. Delays beyond these windows increase rejection rates and necessitate recollection, disrupting workflows.
Chain of custody documentation, including labels with patient identifiers, collection time, and transport conditions, ensures traceability and legal compliance. Couriers must follow regulations from bodies like the International Air Transport Association for dangerous goods, classifying infectious specimens appropriately to prevent exposure during transit.
These fundamentals form the basis for safe, accurate transport, reducing pre-analytical errors that compromise up to 70 percent of laboratory issues.
Best Practices for Packaging and Labeling

Proper packaging and labeling are essential to protect specimens during transit and ensure they are processed correctly upon arrival. Primary containers, such as blood tubes or urine cups, must be tightly sealed and placed in secondary leak-proof packaging with sufficient absorbent material to contain any spills. For air or ground transport of potentially infectious materials, triple packaging systems are standard: a primary receptacle, a secondary watertight container, and a rigid outer package labeled according to regulations.
Labeling must be clear, durable, and include at least two patient identifiers, such as name and date of birth, along with collection date and time, specimen type, and requesting physician. Barcodes enhance tracking and reduce misidentification errors. In one analysis of labeling errors, incomplete data accounted for 0.37 percent of issues, with rates varying by department, reaching 1.02 percent in crossmatch tests.
Temperature indicators or data loggers should accompany sensitive specimens to verify conditions during transit. Insulated containers with cold packs maintain 2 to 8 degrees Celsius for refrigerated items, while dry ice surrounds frozen samples without direct contact to avoid cracking. Packaging must withstand the journey’s stresses, including vibration and pressure changes in air transport.
Couriers trained in biosafety handle Category A or B substances under United Nations recommendations, using proper documentation and spill kits. For routine clinical specimens, non-biohazardous labeling suffices if risks are low.
These practices minimize rejection rates, which can reach 0.79 percent of specimens due to transport issues, ensuring efficient lab operations.
Temperature and Time Requirements for Common Specimens
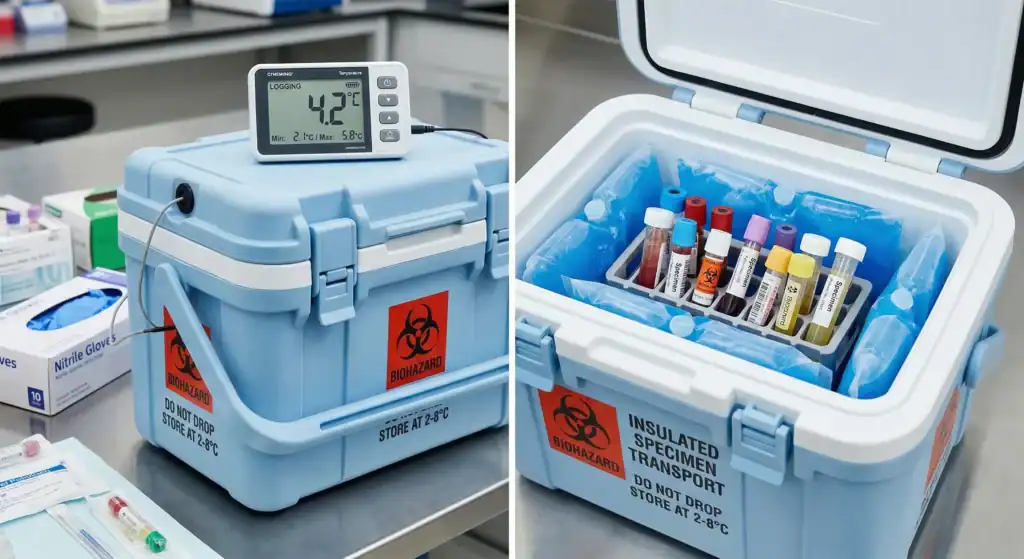
Temperature and time requirements are tailored to specimen type to preserve analyte stability. Blood for chemistry panels should be transported at 2 to 8 degrees Celsius and processed within 2 hours to prevent glycolysis, which lowers glucose by 5 to 7 percent per hour at room temperature. Plasma or serum separated from cells can be refrigerated for up to 24 hours for many tests.
Urine for routine urinalysis tolerates room temperature for short periods but requires refrigeration if delayed beyond 2 hours to prevent bacterial multiplication and pH shifts. For culture, transport within 2 hours at room temperature or 24 hours refrigerated is ideal. Stool specimens for pathogens should be transported promptly or preserved to avoid overgrowth of normal flora.
Swabs for microbiology, such as throat or wound cultures, often use transport media and can be held at room temperature for up to 24 hours. Cerebrospinal fluid requires immediate transport at room temperature, as refrigeration can kill fastidious organisms like Neisseria meningitidis.
Molecular tests for nucleic acids have stricter rules: some RNA-based assays need freezing at minus 70 degrees Celsius if not processed quickly. Guidelines recommend processing within 24 to 48 hours for most specimens, with exceptions for stabilized samples.
Adhering to these parameters reduces invalid results. Studies show transport delays contribute to 10 to 20 percent of pre-analytical rejections, emphasizing the need for timely couriers and monitoring.
Safety Considerations During Specimen Transport
Safety during specimen transport protects couriers, lab staff, and the public from biohazards. All personnel must receive training on handling potentially infectious materials, including use of personal protective equipment like gloves and masks during loading or spills.
Packaging follows triple-layer standards for infectious substances: a primary leak-proof container, secondary packaging with absorbent, and a durable outer box labeled with UN numbers if applicable. Spill response kits must accompany shipments, with protocols for containment and decontamination using appropriate disinfectants like 10 percent bleach.
Regulatory compliance is mandatory. National and international rules, such as those from the Department of Transportation or IATA, classify specimens as Category A (high-risk, like Ebola) or Category B (standard diagnostic specimens). Proper documentation, including shipper declarations, prevents legal issues and ensures safe handling.
In vehicle transport, secure specimens to prevent tipping, and use dedicated coolers separate from food or personal items. For air shipment, dry ice requires venting to avoid pressure buildup. Post-delivery, labs inspect packages for integrity before opening.
These measures have reduced exposure incidents significantly. Training programs have improved compliance rates, lowering risks associated with transport.
Error Rates, Impact of Delays, and Optimization Outcomes

This section presents real data from studies and reports between 2020 and 2025 on specimen transport in clinical laboratories, focusing on error rates, the consequences of delays, and benefits of optimized practices.
Pre-analytical errors dominate laboratory issues, with specimen transport contributing substantially. A 2025 study in Clinical Chemistry and Laboratory Medicine analyzed 37,680,242 billable results from approximately 11,000,000 specimens over 17 months. Total errors affected 0.23 percent of results (2,300 per million) and 0.79 percent of specimens (7,900 per million). Pre-analytical errors comprised 98.4 percent (984,000 per million), with hemolysis as the leading cause at 69.6 percent (696,000 per million). Excluding hemolysis, pre-analytical errors still accounted for 94.6 percent of the remaining issues.
Labeling and identification errors occur at rates of 0.39 to 1.12 per 1,000 specimens, per College of American Pathologists Q-Probe studies. In one multispecialty hospital analysis of 74,279 samples, 0.43 percent were canceled due to identification or labeling errors, representing 10.2 percent of total rejections. Incomplete data caused 0.37 percent of errors, highest in inpatient settings (0.56 percent) compared to emergency (0.24 percent) and outpatient (0.07 percent). Type and screen tests had the highest error rate at 0.88 percent, followed by crossmatch red blood cells at 1.02 percent.
Transport delays exacerbate problems. A 2022 study in South Africa on viral load testing optimization found that shifting courier collection from 13:36 to 14:35 increased patient access to same-day blood draws by 6 to 13 percent. Facilities with later collections achieved 1.14 viral load tests per antiretroviral patient annually versus 1.06 for earlier ones, even after adjusting for facility size (p less than 0.05).
In a 2024 study on laboratory processing delays, each 10-minute increase in urinalysis turnaround extended emergency department length of stay by 15 to 20 minutes (p less than 0.05). Automation in one facility reduced culture turnaround from 48 to 36 hours, enabling earlier antibiotic adjustments and lowering sepsis mortality by 8 percent in 500 intensive care unit patients.
A 2025 Ethiopian proficiency testing analysis showed acceptable performance at 67.6 percent overall, with failures declining from 40.3 percent in 2020 to 20.6 percent in 2022 due to corrective actions, including improved transport protocols. Biosafety training in multiple African studies increased new safety practice implementation from 50 percent to 84 percent.
Optimization yields clear benefits. Real-time tracking and temperature sensors reduce rejection rates, with labs reporting 10 to 20 percent improvements in specimen integrity. In one network, optimized courier timing enhanced access without increasing costs.
These data, from large cohorts, confirm that transport errors affect 0.23 to 0.79 percent of specimens, with pre-analytical issues dominant, and interventions like timely collection or automation cutting delays and improving outcomes.
Challenges and Solutions in Specimen Transport
Challenges in specimen transport include logistical issues in remote areas, where delays exceed 24 hours and increase rejection rates by 10 to 20 percent. Temperature fluctuations during transit cause hemolysis or bacterial overgrowth. Regulatory complexity for infectious specimens adds an administrative burden.
Solutions involve dedicated couriers with temperature-controlled vehicles, real-time GPS and sensor monitoring, and standardized training. Partnerships with professional services ensure compliance. For high-volume labs, pneumatic tube systems within facilities reduce internal transport times to minutes.
Investing in these solutions lowers error rates and supports reliable diagnostics.
Conclusion
Specimen transport in clinical laboratories is essential for maintaining accuracy and safety, with best practices focusing on proper packaging, temperature control, timely delivery, and documentation. Real data show pre-analytical errors at 0.79 percent of specimens, with transport delays contributing to longer stays and higher rejection rates. Optimized systems reduce these issues by 10 to 25 percent and improve patient access and outcomes. Laboratories that prioritize robust transport protocols ensure reliable results, protect personnel, and enhance overall diagnostic quality. As healthcare evolves, continued adherence to standards and innovation in tracking will further strengthen this critical process.