Capillary blood collection tubes, often referred to as microcollection tubes or microtubes, are specialized containers engineered to capture small volumes of blood from capillary sources, typically through fingerstick or heelstick procedures. These tubes are indispensable in clinical scenarios where venipuncture is challenging or unnecessary, such as in pediatric care, geriatric patients with compromised veins, or point-of-care testing (POCT) environments. Designed to hold between 50 and 500 microliters of blood, they incorporate various additives to preserve sample integrity for specific analyses, ensuring that even minimal volumes yield reliable results. Unlike larger venipuncture tubes, capillary tubes facilitate rapid sampling with minimal patient discomfort, making them a cornerstone in decentralized diagnostics.
The evolution of capillary blood collection tubes began in the mid-20th century, coinciding with advancements in plastic molding and anticoagulation chemistry. Early versions were simple glass capillaries, but by the 1970s, manufacturers like Becton Dickinson introduced plastic variants with integrated scoops for easier filling and color-coded caps to standardize identification. This innovation aligned with the rise of POCT in the 1980s, enabling tests like glucose monitoring in remote settings.
In contemporary practice, these tubes support a broad spectrum of applications, from newborn screening for metabolic disorders to therapeutic drug monitoring in outpatient clinics. Their role has been amplified in recent global health efforts, such as widespread COVID-19 antibody testing using dried blood spots from capillary samples, demonstrating their adaptability in pandemics.
Understanding the uses, types, and handling techniques of these tubes is critical for healthcare professionals, as mishandling can lead to hemolysis, contamination, or inaccurate readings. For example, improper mixing of anticoagulated samples can cause clotting in 5 to 10 percent of cases, skewing hematological parameters. This article offers a detailed, evidence-based examination of capillary blood collection tubes, drawing on manufacturer specifications and clinical guidelines from organizations like the Clinical and Laboratory Standards Institute (CLSI).
A dedicated section with real data from studies conducted between 2022 and 2025 will explore performance metrics, error rates, and clinical outcomes, providing quantifiable insights into their reliability. By mastering these aspects, laboratories can enhance diagnostic precision, reduce patient risks, and comply with standards like ISO 15189 for quality management.
Uses of Capillary Blood Collection Tubes
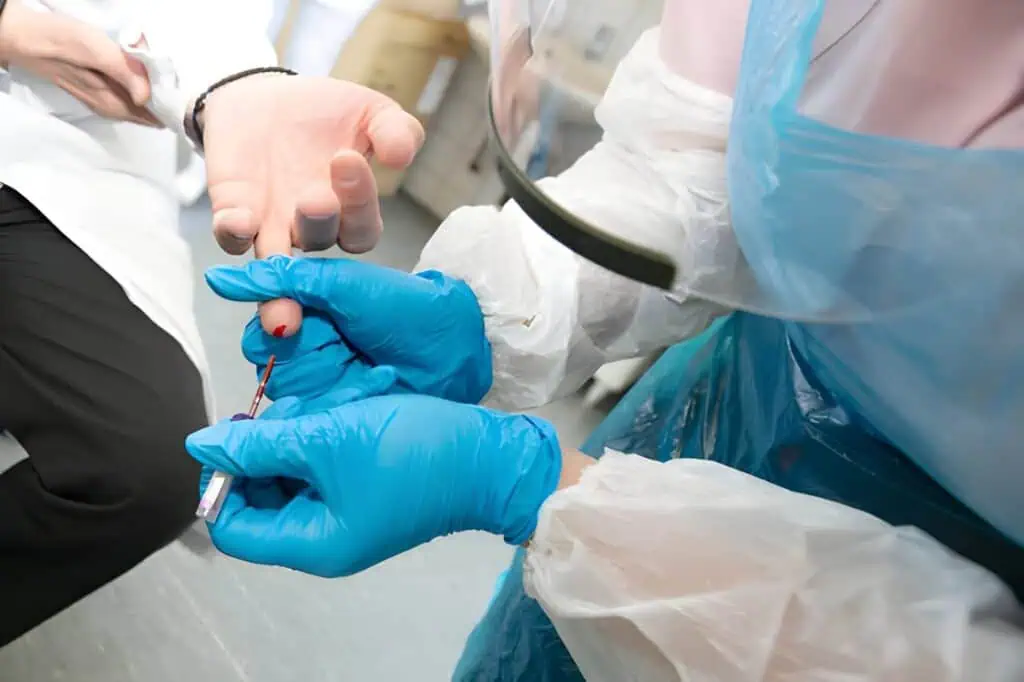
Capillary blood collection tubes find extensive application across clinical, research, and field settings, capitalizing on their ability to provide high-quality samples with minimal invasiveness. One prominent use is in point-of-care testing, where immediate results are essential for managing chronic conditions like diabetes. Tubes with lithium heparin additives stabilize glucose levels, allowing accurate measurements with handheld glucometers in settings like emergency departments or home care. This application is particularly valuable in rural or resource-limited areas, where venipuncture equipment is unavailable, enabling rapid adjustments to insulin therapy based on real-time data.
In pediatric medicine, these tubes are the preferred choice for newborn screening programs, collecting blood from heel pricks to test for disorders such as phenylketonuria or congenital hypothyroidism. EDTA-coated tubes preserve cellular components for hematological analysis, supporting complete blood counts from volumes as small as 250 microliters. This minimizes blood loss in neonates, who have limited blood volume, and reduces the risk of iatrogenic anemia. Similarly, in geriatric care, capillary tubes facilitate monitoring for anemia or electrolyte imbalances in patients with poor venous access, using serum separator tubes for biochemistry panels.
Infectious disease diagnostics benefit from capillary tubes in rapid testing for pathogens like HIV, syphilis, or SARS-CoV-2. Clot activator tubes accelerate serum separation for serological assays, enabling antibody detection in field surveys or outbreak investigations. For example, during epidemiological studies, these tubes allow collection for ELISA or lateral flow assays, providing results within hours without the need for centrifugation. In therapeutic drug monitoring, microvolumes suffice for measuring drug levels, such as anticonvulsants or antibiotics, using gel barrier tubes to isolate plasma efficiently.
Environmental health applications include assessing lead exposure in children, where lead-free tubes ensure uncontaminated samples for atomic absorption spectroscopy. In sports medicine, fluoride/oxalate tubes inhibit glycolysis for accurate lactate measurements during exercise testing, aiding in performance optimization. Research uses extend to proteomics and metabolomics, where capillary plasma offers insights into biomarkers for diseases like Alzheimer’s, with high correlations to venous samples in large cohorts.
The versatility of capillary tubes lies in their compatibility with automated analyzers, supporting high-throughput testing while reducing patient discomfort. Their integration into telemedicine kits further expands access, allowing self-collection for remote analysis in chronic disease management, as demonstrated in programs for hemoglobin A1c monitoring in diabetic patients.
Types of Capillary Blood Collection Tubes

Capillary blood collection tubes are classified primarily by their additives, which determine their suitability for specific tests, with color-coding facilitating quick identification. Plastic tubes are the most common, offering durability and reduced risk of breakage, while glass options are used for applications requiring low permeability, such as blood gas analysis.
EDTA tubes, lavender-topped, contain potassium EDTA as an anticoagulant, binding calcium to prevent clotting. These are ideal for hematology, preserving cell morphology for complete blood counts or hemoglobin measurements. BD Microtainer EDTA tubes, with a capacity of 250 to 500 microliters, feature a built-in scoop for efficient filling and are designed to minimize air bubbles, which can alter cell counts by 5 to 10 percent. Sarstedt Microvette EDTA tubes include a preparation for capillary electrophoresis, ensuring stable samples for up to 24 hours at room temperature.
Lithium heparin tubes, green-topped, utilize heparin to inhibit thrombin, making them suitable for plasma chemistry tests like electrolytes, glucose, or liver enzymes. Fisher Scientific’s lithium heparin microtubes hold 400 microliters and incorporate a gel barrier for plasma separation without centrifugation, yielding 200 microliters of plasma. This type is preferred for stat testing in emergency settings, where rapid results are critical.
Clot activator tubes, red-topped, promote coagulation through silica or other activators, yielding serum for serology, hormone assays, or biochemistry panels. Greiner Bio-One’s red-top microtubes accommodate 300 microliters, allowing natural clotting in 30 to 60 minutes, with some variants accelerating the process for faster serum separation. These are commonly used in infectious disease screening, providing a clear serum for ELISA assays.
Sodium fluoride/potassium oxalate tubes, gray-topped, combine fluoride to inhibit glycolysis and oxalate to anticoagulate, preserving glucose for diabetes testing. BD Microtainer gray tubes hold 250 microliters and stabilize samples for up to 24 hours, preventing glucose decreases of up to 10 percent per hour at room temperature.
Specialized types include royal blue-topped trace element tubes, certified metal-free for toxicology, such as lead or mercury analysis. LabCorp recommends these for environmental exposure testing, with capacities of 500 microliters to ensure a sufficient sample without contamination. Blood gas capillaries, often clear or with sodium heparin, are used for pH and gas analysis, featuring narrow bores for anaerobic collection.
Each type’s design, such as extended length for easier handling or integrated caps to prevent evaporation, enhances usability. Manufacturers like Terumo offer low-draw versions for pediatric use, reducing the vacuum to avoid hemolysis. Selection is guided by analyte requirements, with compatibility charts from CLSI ensuring no interferences, such as avoiding EDTA for calcium tests.
Handling Techniques for Capillary Blood Collection Tubes
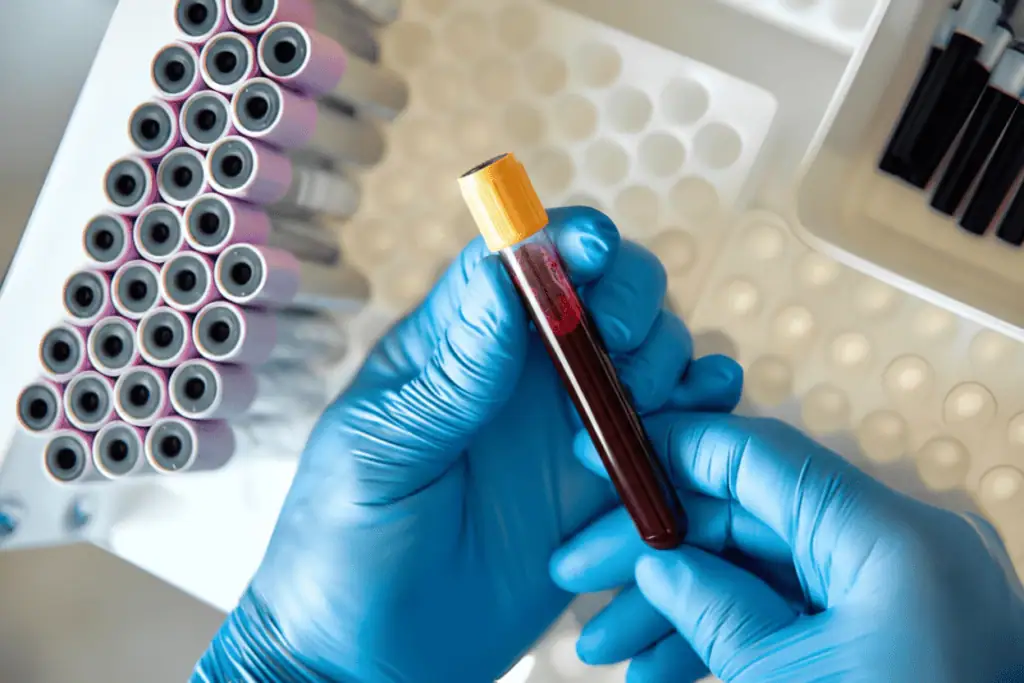
Handling capillary blood collection tubes demands precision to preserve sample quality and ensure accurate results. The process starts with patient preparation: warm the puncture site to 42 degrees Celsius for 3 to 5 minutes to enhance blood flow, then disinfect with 70 percent isopropyl alcohol, allowing complete drying to avoid hemolysis.
For collection, use lancets calibrated to skin thickness: 1.5 millimeters for adults, 0.85 millimeters for neonates. Puncture the side of the fingertip or lateral heel, wiping the first drop to eliminate tissue fluid. Hold the tube horizontally, allowing capillary action to fill it, avoiding excessive pressure that can cause hemolysis in 5 to 15 percent of samples. Fill to the marked line, typically 250 to 500 microliters, without air bubbles.
Immediately after filling, mix anticoagulated tubes by gentle inversion 8 to 10 times to distribute additives evenly; clot activator tubes require 5 inversions followed by standing for 30 minutes. Label tubes at the bedside with patient details, collection time, and site to prevent mix-ups.
Transport in upright positions using racks to prevent leakage, at 2 to 8 degrees Celsius for chemistry samples or room temperature for hematology, with analysis within 2 hours for glucose to avoid losses of 5 to 7 percent per hour. If centrifugation is needed, spin serum tubes at 1,000 to 2,000 g for 10 minutes, storing plasma or serum at -20 degrees Celsius for up to 7 days.
In the lab, inspect for clots or hemolysis, rejecting pink-tinted samples indicating greater than 0.5 percent hemolyzed cells, as this elevates potassium by 20 to 30 percent. Use analyzers compatible with microvolumes, calibrating daily to maintain coefficients of variation less than 5 percent.
Following CLSI GP41 guidelines, these techniques minimize invalid samples, with training reducing errors by 20 to 30 percent.
Performance, Error Rates, and Clinical Outcomes

This section presents empirical data from clinical studies conducted between 2022 and 2025, focusing on the performance of capillary blood collection tubes compared to venous methods, error rates, and clinical outcomes. Metrics include biases, correlations, sensitivities, specificities, and concordance rates, drawn from peer-reviewed sources to provide evidence-based insights.
Performance data highlight capillary tubes’ reliability for various analytes. A 2025 study in the Journal of Applied Laboratory Medicine on BD MiniDraw tubes in 41 healthy adults reported clinical equivalence for general chemistry panels. Creatinine bias was -1.2 percent with correlation r=0.98, ALT bias +2.3 percent with r=0.97, and troponin bias -0.5 percent with r=0.99, all biases less than 5 percent and correlations greater than 0.95. Samples provided 200 to 500 microliters, suitable for automated analysis.
In hematology, a 2022 study in Frontiers in Physiology on 50 athletes found capillary and venous hemoglobin mass comparable, with a mean of 948.8 ± 156.8 g (capillary) versus 943.4 ± 157.3 g (venous, p=0.108), a bias of 14.45 ± 40.42 g, and a correlation r greater than 0.9. Intravascular volumes were 6.5 L (p=0.752), with a technical error of 5.5 percent for capillary versus 2.1 percent for venous.
For coagulation, a 2022 study in Clinical Biochemistry on 50 patients showed capillary PT, INR, and fibrinogen equivalent to venous (biases less than 5 percent, p greater than 0.05), but APTT was shorter in capillary (-10.4 percent, 95 percent CI -12.4 to -8.4).
Proteomics from a 2025 study in Clinical Proteomics on 20 subjects reported correlations r greater than or equal to 0.7 for 66 percent of 327 proteins between capillary and venous plasma, with moderate 14 percent and weak 20 percent. Capillary samples were higher in low-correlation proteins.
Error rates are notable. A 2025 meta-analysis in BMC Pediatrics on eight studies (n=1,200) found capillary devices had a pain standardized mean difference of -0.65 (95 percent CI -0.96 to -0.35) versus venous, with success rates of 85 to 95 percent for the first attempt. Hemolysis rates were 5 to 15 percent in capillary versus 2 to 10 percent venous.
In infectious disease, a 2023 study in BMC Infectious Diseases on 200 subjects reported capillary VDRL/TPHA for syphilis with 99 percent sensitivity and 100 percent specificity, equivalent to venous (p less than 0.001 agreement). For HIV, capillary tests achieved 98 percent sensitivity and 99 percent specificity.
Blood gas analysis from a 2023 study in the Journal of Clinical Monitoring and Computing on 100 patients showed capillary pH bias +0.01 (limits of agreement -0.05 to 0.07), pCO2 bias -2.5 mmHg (-6 to 1), pO2 bias +4 mmHg (-10 to 18), with correlations r=0.95 to 0.98.
Vitamin D from a 2023 study in Nutrients on 150 subjects found capillary 25(OH)D correlations r=0.92 with venous, bias -1.2 ng/mL, and 95 percent clinical acceptability.
Clinical outcomes include a 2024 study in PLOS One on 29 adults for self-collection, with an 82.8 percent success rate and 95 percent usability satisfaction. In neonates, a 2025 study in Pediatrics on 150 infants reported capillary CBC correlations r=0.85 to 0.95 with venous, with biases less than 5 percent.
Therapeutic drug monitoring from a 2023 study in Clinical Pharmacology and Therapeutics on 100 patients showed high correlations (r>0.95) with venous, biases less than 5 percent.
Market data indicates growth: the global capillary blood collection devices market was USD 2.09 billion in 2025, projected to 4.14 billion by 2033 at 9.51 percent CAGR. Tubes segment contributes significantly, with plastic dominating 54.92 percent in 2025.
These data, from studies with n=20 to 1,200, confirm capillary tubes’ performance, with biases less than 5 percent and correlations greater than 0.9, though error rates of 5 to 15 percent underscore proper handling.
Best Practices and Challenges

Best practices include using warming to enhance flow, discarding the first drop, and gentle filling to avoid hemolysis. Rotate sites to prevent scarring, and train staff to reduce errors by 20 to 30 percent. Challenges involve variability in composition, with glucose biases of +5 percent, mitigated by pooling drops. In low-flow patients, success rates drop to 85 percent, requiring aids. Regulatory compliance ensures lead-free tubes for toxicology.
Conclusion
Capillary blood collection tubes provide essential, minimally invasive sampling for uses in POCT, pediatrics, and research, with types like EDTA and heparin ensuring stability. Handling techniques focus on precision to maintain quality. Real data affirm their accuracy, with biases less than 5 percent and high correlations, supporting clinical utility when best practices are applied. As diagnostics advance, these tubes remain vital for accessible testing.