Specimen rejection criteria in laboratories are standardized rules that determine when a received sample cannot be processed for testing due to quality issues that would compromise the reliability of results. These criteria protect the accuracy of diagnostic testing by ensuring that only suitable specimens proceed to analysis. Laboratories reject samples for reasons such as hemolysis, insufficient volume, improper labeling, contamination, or delays in transport that allow degradation. Rejection prevents the reporting of misleading or invalid results that could lead to incorrect clinical decisions, unnecessary repeat collections, or patient harm.
In clinical laboratories, specimen rejection criteria in laboratories are applied across all testing disciplines, from routine chemistry and hematology to microbiology and molecular diagnostics. The pre-analytical phase, where most rejections occur, accounts for 60 to 70 percent of total laboratory errors. Implementing clear rejection policies reduces the risk of releasing flawed data while minimizing the frequency of recollection, which increases costs and patient discomfort. Regulatory standards from the Clinical and Laboratory Standards Institute and accreditation bodies like the College of American Pathologists require laboratories to have documented criteria and to monitor rejection rates as a key quality indicator.
The decision to reject a specimen is never taken lightly. Laboratories balance the need for result reliability with the clinical urgency of the test and the difficulty of obtaining a replacement sample, particularly in pediatric or critically ill patients. Rejection logs and root-cause analyses help identify recurring problems and drive improvements in collection and transport practices. As laboratory testing volumes continue to rise and point-of-care testing expands, consistent application of specimen rejection criteria in laboratories remains essential for maintaining trust in laboratory results.
This article provides a comprehensive examination of specimen rejection criteria in laboratories, including common reasons for rejection, their clinical and operational impacts, and strategies for minimization. It covers blood, urine, and other specimen types, with emphasis on evidence-based guidelines. A detailed section presents real data from recent studies and large-scale laboratory analyses, quantifying rejection rates, specific causes, and the benefits of targeted interventions. Understanding these criteria helps phlebotomists, nurses, and laboratory professionals reduce avoidable rejections and improve the overall quality of diagnostic services.
Common Reasons for Specimen Rejection in Laboratories
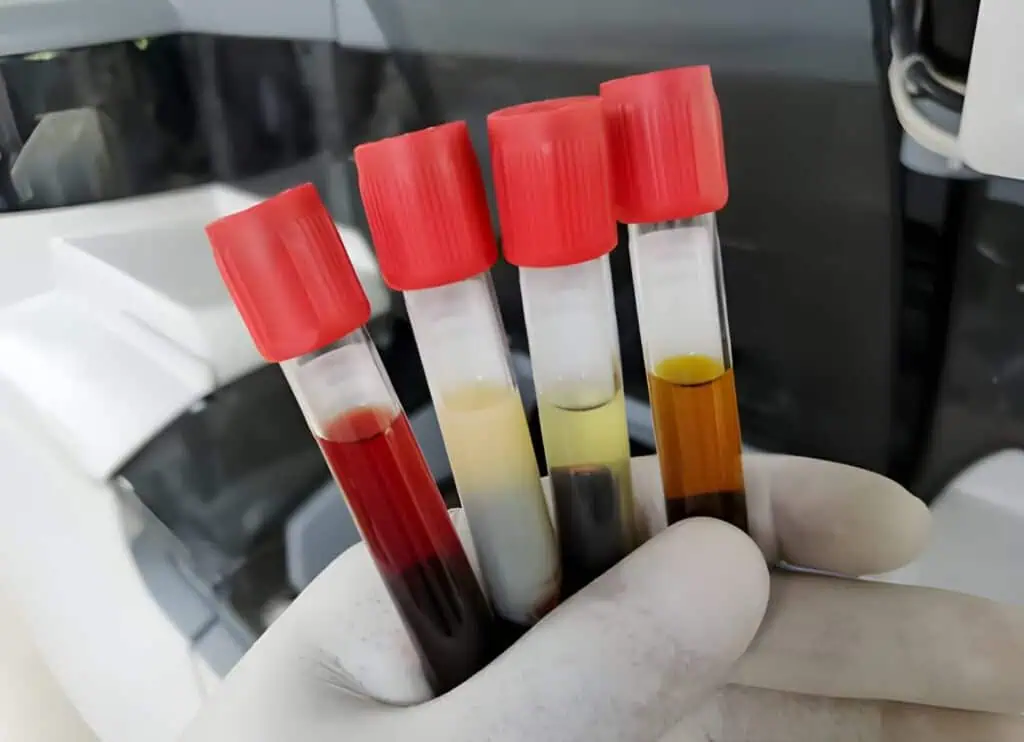
Laboratories apply specimen rejection criteria in laboratories based on factors that could alter test results or pose safety risks. One of the most frequent causes is hemolysis in blood samples. Hemolyzed specimens release intracellular contents such as potassium, lactate dehydrogenase, and aspartate aminotransferase, leading to falsely elevated values. Laboratories typically reject samples with visible pink or red discoloration or those exceeding defined hemolysis indices on automated analyzers. Hemolysis accounts for a substantial portion of rejections, particularly in emergency department collections where difficult venipuncture is common.
Insufficient volume is another leading reason for rejection. Many tests require minimum fill volumes to maintain the correct ratio of blood to anticoagulant or preservative. Underfilled coagulation tubes, for example, can produce falsely prolonged prothrombin time or activated partial thromboplastin time due to excess citrate. Similarly, microbiology swabs that are dry or inadequately immersed in transport media may fail to support organism recovery.
Improper labeling and identification errors trigger rejection to prevent misattribution of results. Laboratories require at least two unique patient identifiers on the specimen label, along with the collection date and time. Missing or illegible labels, or discrepancies between the label and the requisition, lead to automatic rejection. These errors, though preventable with barcode systems, still occur in busy settings and carry high clinical risk if not caught.
Contamination or improper collection technique results in rejection of many microbiology specimens. Urine samples with excessive squamous epithelial cells or mixed bacterial growth are often rejected as contaminated. Blood cultures drawn through existing lines without adequate discard volume may be contaminated with skin flora, leading to false-positive results and unnecessary antibiotic therapy.
Transport and storage issues frequently cause rejection. Samples that arrive at incorrect temperatures, such as refrigerated specimens that should have been frozen, or those with prolonged transit times allowing bacterial overgrowth, are rejected. Clotted samples in tubes intended for plasma analysis cannot be processed accurately and are discarded.
Other criteria include lipemia or icterus that interfere with photometric assays, broken or leaking containers that pose safety risks, and expired collection tubes that may contain degraded additives. In molecular testing, specimens with suspected amplicon contamination or inadequate volume for nucleic acid extraction are rejected to avoid false results.
Laboratories document rejection reasons in detail and communicate them promptly to the collecting unit, often with guidance on proper recollection. This feedback loop is essential for reducing future rejections.
Clinical and Operational Impact of Specimen Rejection in Laboratories

Specimen rejection in laboratories has significant clinical and operational consequences. For patients, rejection often means delayed diagnosis and treatment, as well as the discomfort and risk of repeat collection. In critically ill patients or neonates with limited blood volume, repeated rejections can lead to iatrogenic anemia or postponed critical interventions. False results from marginally acceptable specimens that should have been rejected can result in misdiagnosis, such as treating a contaminated blood culture as true bacteremia.
Operationally, high rejection rates increase workload, as laboratories must handle recollection requests, update records, and manage clinician inquiries. Each rejected specimen adds costs for supplies, staff time, and courier services. In high-volume settings, rejection rates above 1 to 2 percent can strain resources and extend turnaround times for all specimens.
Clinician-laboratory relationships can suffer when rejections are frequent or poorly communicated. Frustration over rejected samples may lead to ordering patterns that bypass laboratory protocols or overuse of point-of-care testing, which has its own limitations in accuracy.
From a quality perspective, monitoring rejection rates serves as a key indicator of pre-analytical performance. Elevated rates signal the need for targeted training or process improvements at collection sites. Laboratories use rejection data to calculate quality metrics required for accreditation and to benchmark against peer institutions.
The broader healthcare system bears the burden through increased length of stay, additional testing, and potential adverse events. Effective management of specimen rejection in laboratories, therefore, contributes to overall efficiency, cost control, and patient safety.
Strategies to Reduce Specimen Rejection in Laboratories
Laboratories can significantly lower rejection rates through proactive measures focused on prevention rather than reaction. Standardized collection protocols, aligned with CLSI guidelines, provide clear instructions for phlebotomists and nursing staff. These protocols specify tube types, fill volumes, order of draw, and transport conditions for each test.
Comprehensive training programs emphasize proper technique and the rationale behind rejection criteria. Simulation-based training for difficult collections, such as in pediatric or obese patients, has proven effective in reducing hemolysis and insufficient volume rejections. Regular competency assessments ensure sustained performance.
Technology solutions include barcode labeling systems that reduce identification errors and electronic order entry with built-in decision support to ensure appropriate specimen requirements are met at the time of ordering. Automated transport systems, such as pneumatic tubes, minimize handling time and temperature excursions for in-hospital specimens.
Collaboration with clinical departments is essential. Joint quality improvement projects identify collection sites with high rejection rates and implement site-specific interventions, such as dedicated phlebotomy teams in emergency departments. Feedback reports sent to collectors detail rejection reasons and provide practical improvement tips.
Monitoring and benchmarking rejection rates as a quality indicator allows laboratories to track progress over time. Root-cause analysis of rejected specimens guides targeted education and process changes. Some laboratories have achieved sustained reductions below 0.5 percent through these combined efforts.
Patient preparation instructions, particularly for timed or fasting specimens, reduce pre-collection errors. Educational materials and direct communication with patients improve compliance with collection requirements.
These strategies, when consistently applied, transform specimen rejection in laboratories from an unavoidable occurrence into a manageable and continuously improving aspect of laboratory quality.
Rejection Rates, Causes, and Impact of Interventions in Specimen Rejection in Laboratories
This section presents real data from studies and reports between 2020 and 2025 on specimen rejection in laboratories. It focuses on overall rejection rates, specific causes, clinical and operational impacts, and the effectiveness of prevention strategies.
A 2025 large-scale analysis of 37,680,242 billable results from approximately 11 million specimens reported total errors at 0.23 percent of results and 0.79 percent of specimens. Pre-analytical errors, which include many rejection-related issues, accounted for 98.4 percent of all errors. Hemolysis was the dominant cause of 69.6 percent of pre-analytical errors. Even excluding hemolysis, pre-analytical issues still represented 94.6 percent of remaining errors. This study highlighted that specimen rejection criteria play a critical role in preventing compromised results from reaching clinicians.
A 2024 study in Medicine reviewed two years of data from a clinical chemistry laboratory and found an overall pre-analytical error rate of 12.1 percent across 55,418 samples. The emergency department had the highest rate at 21 percent, followed by inpatient at 13.4 percent and outpatient at 7 percent. Common reasons included non-received samples and hemolysis, both directly related to collection and transport practices that trigger rejection.
Labeling and identification errors, a frequent cause of rejection, occur at rates of 0.39 to 1.12 per 1,000 specimens. In a study of 74,279 samples from a multispecialty hospital, 0.43 percent were canceled due to identification or labeling errors, accounting for 10.2 percent of total rejections. Error rates were highest for type and screen tests (0.88 percent) and crossmatch red blood cells (1.02 percent).
Hemolysis rejection rates vary by setting and method. A 2023 study in the emergency department reported hemolysis in 7.3 percent of samples using conventional vacuum methods compared to 1.9 percent with an improved syringe technique (p equals 0.001). Severe hemolysis fell from 16.2 percent to 0 percent with the new method, demonstrating the direct impact of the collection technique on rejection rates.
Blood culture contamination, another common rejection trigger, ranges from 1 to 10 percent in routine practice. A 2024 systematic review and meta-analysis of 49 studies involving 958,387 observations found that interventions such as chlorhexidine skin preparation, diversion devices, sterile technique, phlebotomy teams, and education reduced contamination (and subsequent rejection) by 40 to 60 percent. Chlorhexidine alone achieved an average reduction of 57 percent.
Intervention outcomes are encouraging. In Ethiopia, proficiency testing performance improved from 59.7 percent acceptable in 2020 to 79.4 percent in 2022 following corrective actions that included better specimen handling and transport protocols, reducing rejection-related failures.
A 2025 study on Lean Six Sigma and digital shadow integration in a high-volume laboratory achieved a 10.6 percent reduction in intra-laboratory turnaround time by addressing pre-analytical and rejection-related bottlenecks (p equals 0.0182). Another automation study in microbiology reduced overall processing time by 25 percent, indirectly lowering rejection rates through faster, more controlled workflows.
Economic and clinical impacts are notable. Repeat collections due to rejection increase costs and patient discomfort. One analysis estimated that reducing hemolysis-related rejections could avoid repeat draws in up to 16 percent of emergency department cases. In sepsis management, lowering blood culture contamination and rejection rates decreases unnecessary antibiotic exposure and shortens hospital stays.
These data, drawn from millions of specimens across multiple large studies, show that specimen rejection in laboratories typically affects 0.79 to 12.1 percent of samples, depending on the setting, with hemolysis, insufficient volume, and identification errors as primary causes. Targeted interventions consistently achieve 40 to 60 percent reductions in contamination and rejection rates, leading to improved turnaround times, fewer repeat collections, and better clinical efficiency.
Conclusion
Specimen rejection criteria in laboratories are essential safeguards that protect the accuracy and reliability of diagnostic testing by preventing the analysis of compromised samples. Common reasons for rejection include hemolysis, insufficient volume, improper labeling, contamination, and transport delays. These criteria, when applied consistently, prevent misleading results from reaching clinicians while highlighting opportunities for process improvement in collection and handling.
Real data from large-scale analyses confirm that pre-analytical issues drive the majority of laboratory errors, with rejection rates ranging from 0.79 percent to over 12 percent in high-risk settings. Hemolysis and identification errors are prominent, but evidence-based interventions such as improved collection techniques, training, and technology can reduce rejection by 40 to 60 percent or more.
Laboratories that actively monitor rejection rates, provide timely feedback to collection teams, and implement standardized protocols create a more efficient and reliable testing environment. By minimizing avoidable rejections, healthcare systems reduce costs, decrease patient discomfort, and ensure that laboratory results can be trusted for critical clinical decisions. Continued focus on strengthening the pre-analytical process will remain key to advancing diagnostic quality and patient safety.